This folder contains 31 pages of memos, reports, and technical notes about the Light Benzol Absorber (LBA) system spanning from 1932 through 1964. Together, these documents trace more than three decades of experimentation, troubleshooting, and incremental improvements in the recovery of benzol and other light oils from coke oven gas. They offer a rare window into the day-to-day reality of by-product plant operations—where efficiency wasn’t just a goal but a necessity. This folder was recovered from the plant on March 6, 2021. For those interested in exploring the complete file, the full PDF can be downloaded below.

Laura Kessler:
This memo is almost a century old. What was the purpose of this document?
Frank Malloy:
At its heart, it’s an internal report evaluating two methods of recovering benzol—an aromatic hydrocarbon by-product—from coke oven gas. In 1932, by-product recovery was crucial. Plants were desperate for any revenue to offset the economic devastation of the Great Depression. Capturing benzol wasn’t just about efficiency—it was about survival.
L.K.:
What exactly is a Light Benzol Absorber, or LBA?
F.M.:
The LBA is a piece of equipment that removes “light oils” like benzol, toluol, and xylol from the raw gas coming off the coke ovens. Imagine it as a tall column where hot gas rises while a special absorption oil trickles down. The benzol vapors dissolve into the oil, cleaning the gas and capturing valuable chemicals. The “light” in the name refers to the relatively low boiling point of these compounds compared to heavier tars.
L.K.:
The memo compares two operating methods. What are they?
F.M.:
The first method introduces debenzolized oil—oil that has already been cleaned of benzol—directly into the scrubber. This gives you maximum absorption capacity.
The second method recirculates oil that’s partially saturated, maintaining it within a target range of about 3% benzol. In theory, that reduces the need to regenerate fresh oil constantly.
L.K.:
Which method did they decide was better?
F.M.:
Using freshly debenzolized oil proved superior. It resulted in:
- Higher scrubbing efficiency—more benzol recovered
- Less steam consumption, since you don’t have to reboil partially loaded oil as often
- A higher yield of light oil per ton of coal processed
L.K.:
Why did steam consumption matter so much?
F.M.:
Steam was the hidden cost in by-product recovery. You needed it to regenerate the absorption oil by heating it to release the absorbed benzol. Less steam meant:
- Lower fuel costs, since you were burning less coal or coke oven gas to produce steam
- Less wear and tear on equipment, because heating systems were prone to leaks and scale buildup
During the Depression, those savings could be the difference between staying open or shutting down.
L.K.:
There’s a very formal tone to this memo. Was that typical?
F.M.:
Absolutely. Even internal plant correspondence from this era was precise and respectful. It reflects a culture where technical communication carried real authority. Notice the phrasing:
“The writer would like to call to your attention…”
Even a critique was wrapped in politeness.
L.K.:
What does this document tell us about the people who worked at Acme Coke?
F.M.:
It shows they were methodical and innovative. Even amid economic crisis, they were testing process adjustments and documenting the results carefully. This memo captures the mindset that made the coke industry a powerhouse: relentless experimentation, practical measurement, and an unshakable focus on yield.
L.K.:
Why does this matter today?
F.M.:
Because it’s a rare window into industrial life when chemical recovery was still evolving. Most people picture coke production as a dirty, brute-force process. But here you see the reality: careful optimization, detailed record-keeping, and a constant search for efficiency. In a sense, this memo is an artifact of early process engineering as we know it.

Laura Kessler:
Frank, these four pages are dense—can you give us a high-level overview?
F.M.:
Absolutely. These are daily operating records from late December 1931 into early January 1932, when the plant was experimenting with how to best scrub benzol and light oils from coke oven gas. You have:
- A narrative timeline describing which absorber was in use each day and how the oil feeds and steam were adjusted.
- A table tracking steam consumption.
- A table tracking volatile matter, stripping efficiency, and oil yields.
- A table recording fractional distillation data to see exactly what the light oil contained.
Put together, it’s a snapshot of methodical process experimentation.
L.K.:
Let’s start with the first page—the narrative log. What were they doing?
F.M.:
They were switching between two main operating strategies:
- Using fully debenzolized oil (fresh oil with almost no benzol in it) to maximize absorption capacity.
- Recirculating partially loaded oil to reduce steam consumption.
They also experimented with operating two absorbers in series—meaning gas passed through one absorber, then the second, for deeper cleaning. You see entries like:
“The #1 and #2 L.B.A.’s were in operation in series…”
That was all about finding the sweet spot between steam cost and absorption performance.
L.K.:
The second page shows steam consumption figures. How should we read this?
F.M.:
This table shows, day by day:
- Pounds of steam consumed over 24 hours.
- How much steam was needed per gallon of oil produced and per gallon of wash oil circulated.
- Gallons of “Straw Oil” and “Light Oil” output.
“Straw Oil” was the wash oil circulating through the scrubbers. “Light Oil” was the product containing benzol and toluol fractions.
Notice how on days when they recirculated partially loaded oil, total steam use dropped, but light oil yields often decreased or became inconsistent. That was the trade-off they were trying to quantify.
L.K.:
What do the notes like “Recirculating on #2 L.B.A. account of low city gas” mean?
F.M.:
Sometimes city gas supply—fuel for producing steam—was restricted. Recirculating oil was a way to operate with less steam available, even if it compromised scrubbing efficiency. That note is essentially an operator’s explanation for why they had to change tactics on those days.
L.K.:
The third page is labeled “Operating Data.” What stands out here?
F.M.:
This page is crucial. It records:
- Volatile Matter % in the oil—a measure of how much benzol and light hydrocarbons were left after stripping.
- Stripping Efficiency—how effectively the still removed absorbed compounds.
- Gallons of light oil per day and per ton of coal processed.
- Temperatures of the oil entering the still and the absorber.
This shows how well the process was cleaning the oil and how much product they were recovering. You can see the struggle to keep volatile matter below 2%—some days they hit it, others they didn’t. That fluctuation was a sign the process wasn’t fully optimized yet.
L.K.:
Finally, the fourth page lists distillation percentages and naphthalene content. What’s the importance of this?
F.M.:
This is a chemical fingerprint of the light oil:
- Temperature of first drop tells you how volatile the front end was.
- The columns with “% off at 100° / 120° / 150° / 200°” are fractional distillation cuts. Higher percentages mean more low-boiling fractions.
- Dry point is the final boiling temperature—an indicator of the oil’s heaviness.
- Naphthalene grains per 100 cu ft measures how much naphthalene stayed in the cleaned gas—important for preventing condensation and clogging in distribution lines.
Together, this data told the chemists whether the recovered oil met quality specs for sale or blending.
L.K.:
What do these four pages show about how the plant operated?
F.M.:
They show a high degree of discipline and experimentation. Even in 1932, they were meticulously measuring every pound of steam, every gallon of oil, and every fraction of yield. They were trying to find the most economical way to run the absorbers without compromising product quality. It’s impressive—even by today’s standards.
L.K.:
Why do you think they documented this level of detail?
F.M.:
Two reasons. First, benzol recovery was a major revenue stream. Every gallon mattered. Second, this was still an evolving science. There was no textbook that told you exactly how to balance recirculation, steam economy, and purity. They had to test, log, and adjust day by day.
L.K.:
If you were explaining this to someone who’s never seen a coke plant, how would you sum it up?
F.M.:
I’d say this: Imagine you’re running a giant chemical sponge that soaks up valuable hydrocarbons. Every hour, you have to decide: do I wring it out completely with a lot of steam? Or let it keep working a little dirty to save fuel? These records are the operators’ playbook for answering that question in the middle of a Chicago winter, almost a century ago.
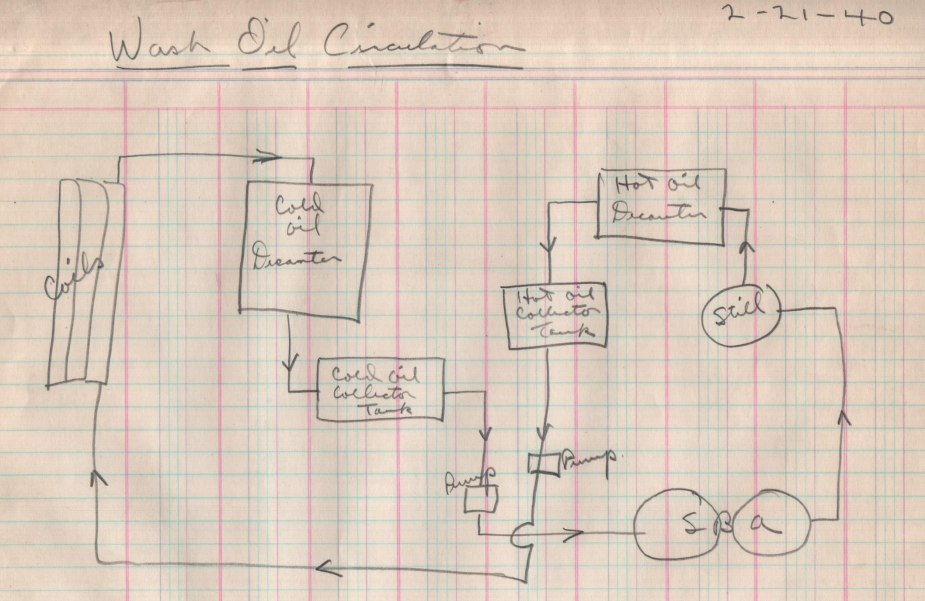
Laura Kessler:
Frank, this document reads more like a lab procedure manual than a typical plant memo. Why is that?
F.M.:
That’s a keen observation. This is a formal specification issued by Koppers, who were leaders in coke by-product engineering. Rather than describing daily operations, it sets out the minimum quality standards wash oil had to meet before it was even brought into the plant. Think of it as the playbook to ensure predictable absorber performance—because if the oil’s viscosity or volatility was off-spec, everything downstream could fail.
L.K.:
Let’s step through each section. The first one covers specific gravity. Why did that matter?
F.M.:
Specific gravity determined how dense the wash oil was—too heavy, and it wouldn’t circulate or atomize properly in the absorber towers. Too light, and it could evaporate excessively or lose absorption capacity. They required it to be no higher than 0.875 at 15.5 °C, which is typical for a midweight petroleum solvent.
L.K.:
Section two is about viscosity. They specify two temperatures. What’s the reason?
F.M.:
Viscosity controlled how well the oil could be sprayed, recirculated, and separated from gas. They tested at 38 °C (roughly 100 °F) to simulate warm operation and 4 °C (near refrigeration) to see if it would thicken in winter. The limits—69 seconds and 150 seconds in a Saybolt Universal Viscosimeter—ensured it was fluid enough for pumping year-round.
L.K.:
Section three describes an emulsification test. Why were they so concerned with separation?
F.M.:
Because if the oil emulsified—formed a stable mix with water—it would create sludge, foul the stills, and ruin efficiency. The test was quite rigorous: shake the oil and water for 20 minutes, then demand 95% separation in just 10 minutes. That was to guarantee smooth decanting and avoid fouling.
L.K.:
Section four talks about boiling range, with very tight parameters. What was the goal there?
F.M.:
This was about controlling volatility. If too much of the oil distilled below 300 °C, it would vaporize in the absorber and carry over into the gas lines—creating contamination or losses. Conversely, if too much remained above 370 °C, it was too heavy to strip clean in the stills. They wanted a sweet spot: stable but volatile enough to pick up benzol.
L.K.:
Section five is the decomposition test. That sounds complicated—can you explain it?
F.M.:
Absolutely. They essentially baked a sealed flask of the oil at 140–145 °C for 120 hours to simulate prolonged heating in operation. Then they washed out residues with petroleum ether and weighed what remained. The goal was to confirm that no more than 0.10% by weight was left as tar or sludge. This told them if the oil was prone to carbonizing in the stills.
L.K.:
The document mentions a distillation apparatus drawing. Is that the schematic we have dated 1940?
F.M.:
Almost certainly. The diagram titled “Wash Oil Circulation” shows the process steps: coils, decanters, pumps, still, and absorbers. While it’s dated 1940—seven years after the spec—it clearly depicts how the oil moved through the system the specs were designed to support.
L.K.:
There’s no author on that drawing, but the handwriting is very similar to K.J. Purcell’s notes. Do you think he drew it?
F.M.:
Based on the lettering, format, and language, I’d say it’s highly probable. Purcell appears across this folder as a kind of in-house chemist or process scientist. He was known to have worked at the Riverdale facility well into the late 1950s and often documented lab protocols in this meticulous style.
L.K.:
How does this specification fit into the bigger picture of coke oven operations?
F.M.:
It’s a reminder that even in the 1930s, success wasn’t just about big machines or brute force—it depended on chemistry. Operators needed to know their wash oil wouldn’t break down or foul the equipment. Without reliable standards like this, production losses could be enormous.
L.K.:
If you had to sum up why this document mattered, what would you say?
F.M.:
I’d say it’s a declaration of precision in an industry often thought of as crude. It’s a statement that even a solvent—something most people never see—had to meet exacting thresholds to keep the entire by-product system working.
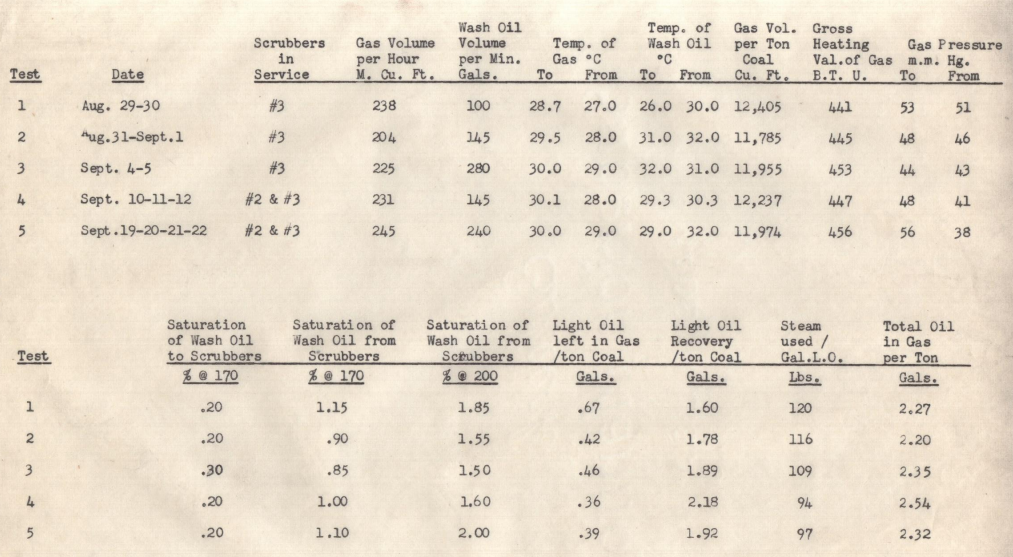
Laura Kessler:
Frank, this memo from Fred Hagedorn and Reg Chamberlin lays out a pretty detailed series of tests. Before we get into the specifics, can you tell me what prompted them to conduct this study at all? Was something going wrong with the light oil recovery?
Frank Malloy:
That’s a great starting point. From everything I’ve seen, nothing was exactly “going wrong,” but they knew the process was inefficient—particularly in steam usage. Remember, steam was a huge cost driver in running the absorbers and regenerating the wash oil.
Fred Hagedorn, who was the plant superintendent, and Reg Chamberlin, his assistant, were both pretty forward-looking. This study was about seeing how much they could push the process to recover more light oil while burning less steam. Even small gains translated into real dollars over time.
L.K.:
I see. Looking at Test 1, it seems like their baseline operation was only using the #3 scrubber. Why would they have been relying on that single vessel, instead of using both scrubbers?
F.M.:
Good observation. Using only the #3 scrubber was simpler—it meant fewer pumps running and less complicated flow control. In a lot of older plants, that was the default: run one absorber at high enough circulation to do most of the scrubbing. But the downside was exactly what you see in Test 1: they were leaving 0.67 gallons of light oil per ton of coal in the gas and consuming 120 pounds of steam per gallon recovered. That was an expensive combination.
L.K.:
Then in Test 2, they increased the wash oil circulation to 1.5 gallons per square foot of grid area. That seems like a big jump in flow. Did that immediately pay off?
F.M.:
Yes and no. From the data, you can see the residual oil in gas dropped significantly—to 0.42 gallons per ton coal—so scrubbing efficiency improved. But they hadn’t yet tackled steam usage; that only came later. So while more oil was recovered, the cost in steam per gallon didn’t change much in Test 2.
L.K.:
Test 3 increased the circulation even more, up to 280 gallons per minute. But the efficiency didn’t improve, did it?
F.M.:
That’s exactly right. You have to look carefully at how these absorbers work. Just pushing more wash oil through doesn’t automatically mean more oil is recovered—especially if you don’t expand the contact area or adjust gas velocity. So in Test 3, they hit diminishing returns on scrubbing efficiency. But they did start improving steam economy—down to 109 pounds per gallon. That probably came from recirculating part of the oil internally and returning only the more saturated fraction to the still.
L.K.:
In Test 4, they switched to using both scrubbers in a counter-current arrangement. That’s when results really improved. Why was that configuration so much better?
F.M.:
You nailed it—counter-current scrubbing is always more efficient because it maximizes the driving force for mass transfer. The cleanest oil contacts the cleanest gas last, so you get better pickup of residual vapors. In Test 4, that configuration cut the residual oil in gas down to 0.36 gallons per ton, while also reducing steam usage to 94 pounds per gallon—their best result so far.
L.K.:
They called Test 5 “approaching the ideal mode of operation,” but the results look pretty similar to Test 4. What were they trying to refine?
F.M.:
They were fine-tuning the balance between re-circulation and fresh circulation to the still. In Test 5, they fed 240 gallons per minute to the large scrubber and only removed 100 gallons to the smaller scrubber and the still. The idea was to maintain the same efficiency while slowly reducing steam usage over time. You see they started a plan to cut steam 500 pounds per hour every second day, watching for signs of rising saturation in the wash oil. That’s how you figure out the absolute minimum steam you can get away with.
L.K.:
Overall, what do you think this testing campaign achieved for the plant?
F.M.:
They accomplished two big things. First, they raised light oil recovery by about a quarter gallon per ton coal, which is significant. Second, they shaved 20% off steam consumption. For an operation running day and night, that’s a major cost reduction. And keep in mind, Fred Hagedorn and Reg Chamberlin did this through methodical, data-driven work—not guesswork. It’s a good example of mid-century industrial process optimization.
L.K.:
One last question—there’s no signature on this memo. Was that typical?
F.M.:
In those days, yes. Internal reports often just listed the authors at the top, in this case Fred and Reg, without a formal sign-off. Everyone knew who was responsible. It was an efficient system, though it looks a little informal by today’s standards.

L.K.:
Frank, these handwritten pages are remarkable. Just to get oriented, can you tell me what Purcell and Klopsch were trying to accomplish with all these fuel oil blend tests?
F.M.:
Absolutely. You’re looking at a methodical attempt to create a fuel blend that matched or improved upon the heating properties of the oil they were using to warm coal cars, especially in winter.
You have to remember, this was before widespread natural gas service in a lot of industrial applications, so fuel oil was critical. But different sources—Standard Oil, kerosene from the oil house, Forest Park blends—had different viscosities and burning characteristics.
So Purcell, who signed this document, and Klopsch, who likely contributed, were systematically mixing proportions of kerosene, gasoline, and heavier oils to see what flowed, ignited, and burned most like their standard fuel oil.
L.K.:
I see. On the first page, they compared several samples and measured specific gravity and viscosity. Why were those properties so important?
F.M.:
Specific gravity tells you how dense the fuel is—important for both metering it and predicting energy content. Viscosity affects how it flows through burners and atomizers.
If you try to pump a heavier oil through nozzles sized for kerosene, you get poor atomization—big droplets that don’t burn well. Conversely, a fuel that’s too light can flash or burn too hot. So they were measuring these properties to confirm whether any blend would handle the same way in their equipment.
L.K.:
They noted “27 sec flow at 4 inches” and “33 sec at 4 inches.” What does that refer to?
F.M.:
That’s a flow test using an orifice or a calibrated viscometer tube—essentially, they time how long it takes a set quantity of fuel to drain through an opening under gravity. The “4 inches” likely refers to the head height or the diameter of the opening.
It’s an old but reliable way to measure relative viscosity, especially in field conditions or small labs.
L.K.:
The second page has all these blend percentages—25% kerosene, 50%, 60%, and so on. What were they looking for in those trials?
F.M.:
They were trying to find the sweet spot where the blend was thin enough to flow and atomize properly but still retained enough density and energy content to burn at the desired temperature.
If you look closely, as they added more kerosene, the final temperatures often fell—like in the 40% and 50% kerosene blends compared to lower percentages. They were cross-referencing specific gravity, temperature, and flow times to see which blend performed closest to their standard oil.
L.K.:
On the third page, it gets even more complex—gasoline, “Stanlin,” light oil, middle burner fuel. Why so many ingredients?
F.M.:
That was experimentation to fine-tune combustion. Each component contributed something:
- Gasoline thinned the blend and boosted volatility.
- Stanlin—probably a proprietary fuel oil—added density.
- Light oil and middle burner fractions affected burn temperature.
It was like creating a recipe: they were adjusting for flow, ignition, stability, and safety.
In the absence of modern analytical equipment, blending in controlled proportions and observing the results was their best method.
L.K.:
The last page shows some blends with no specific gravity or temperatures listed. Do you think those were just proposed formulas, or did they test them?
F.M.:
My guess is those were either in-progress tests or planned blends they hadn’t finished measuring. Sometimes you see partial records like this when a chemist is recording a sequence of ideas, and measurements came later.
It’s also possible the readings were taken but noted elsewhere, or they ran short on time or sample quantities. You can see the work was meticulous, but it was still an industrial setting—not a university lab.
L.K.:
At the bottom of the first page, Purcell wrote that the 50/50 kerosene blend “compares favorably with the oil used to present for heating coal cars.” Would you say that was the main conclusion?
F.M.:
Yes, exactly. The 50/50 blend had nearly identical flow and burning behavior to their standard heating oil. That was probably the preferred formula they were ready to scale up.
But the other blends gave them options—if kerosene or gasoline supply fluctuated, they knew how to adapt. That flexibility was critical for keeping production moving during fuel shortages or price spikes.
L.K.:
This feels like such a hands-on process. Do you think these tests were common practice at the time?
F.M.:
Absolutely. This was classic mid-century industrial chemistry—careful, empirical blending, measured in simple but effective ways.
You didn’t have digital viscometers or chromatographs. You had a good scale, a calibrated orifice, and decades of experience. Purcell and Klopsch were doing exactly what any plant chemist and operations lead would have done: test, document, adjust.
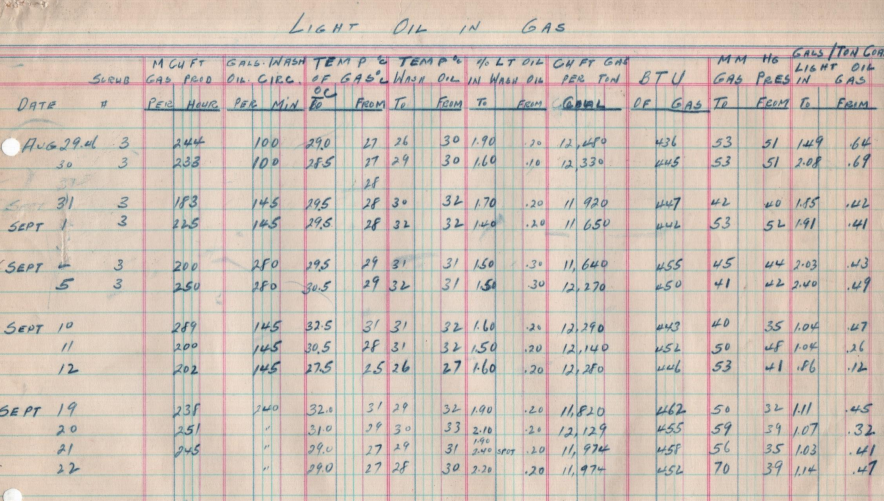
L.K.:
Frank, I noticed something unusual here. This 1949 note from Purcell attaches what looks like the same scrubbing efficiency data I saw in an earlier table—just recopied more neatly. Why do you think they kept both versions?
F.M.:
That’s a sharp catch. You have to remember—this was long before any kind of digital archiving. Everything was filed by hand.
Most companies followed a principle of caution: never throw anything out if you weren’t sure it was redundant. So if someone recopied the table—maybe to clean it up for a report—it often ended up back in the same folder. And nobody wanted to risk discarding the original, just in case there was a note or correction buried in it.
L.K.:
It feels almost paradoxical—so meticulous in measurement, but not always precise about organizing the paperwork.
F.M.:
That’s exactly right. Their recordkeeping was as good as it could be for the time—typed indexes, carbon copies, ledgers. But inevitably, duplicates slipped in.
You can also see their priorities: preserving the data mattered more than streamlining the files. If a superintendent wanted to look back ten years later, they wanted to be certain nothing was missing.
L.K.:
I also noticed Purcell still signed his name, even on an informal note about old records. Was that just his way?
F.M.:
It was partly habit, partly professionalism. Even when you were just forwarding something, you put your name on it. It was a way to say, I’ve checked this; you can rely on it.
It’s one of the small ways you see their respect for each other—and for the integrity of the work itself.
L.K.:
Frank, I’m struck by this exchange about Deep Rock Absorption Oils. It feels like the plant was approached by a very persistent salesman. What was going on here?
F.M.:
You’re right on target—this was Charles Novak, representing the Deep Rock Division of Kerr-McGee. In the 1950s, Deep Rock was trying to expand market share in the Midwest, and absorbent oil was an important commodity for coke plants.
Novak was doing what good salespeople have always done: getting his product spec sheets in front of decision makers, both at the plant in Chicago and the corporate office in Cleveland. Even if they didn’t switch suppliers immediately, he made sure his name and his oil were on file.
L.K.:
I noticed that in the memos, Chamberlin says they were satisfied with their current material. Why wouldn’t they consider switching if Deep Rock offered comparable specifications?
F.M.:
In operations like this, there was a lot of inertia in purchasing. If your current supplier was reliable, and your absorption oil performed consistently, you had every incentive to stick with them. Changing meant new lab tests, possible unexpected behavior in the scrubbers, and the risk of operational headaches.
Chamberlin was being diplomatic: essentially saying, We’re not in a hurry to make a change, but we’ll keep your information on file.
L.K.:
Page three of this packet has a testing procedure that looks older than the rest. Do you think that was originally an Interlake document?
F.M.:
That’s a sharp observation. It might have started as an external spec—maybe something developed by a supplier or a trade association—and over time, Interlake adopted it as their standard method.
This was common. In an era before standardized ASTM methods were universally adopted, each plant accumulated its own reference procedures. Once something worked, it stuck around, sometimes for decades.
L.K.:
I also noticed the references to “olefins.” Why were they so concerned about olefins in their absorbent oil?
F.M.:
Olefins—what chemists now call alkenes—are more reactive than saturated hydrocarbons. In an absorbent oil, they could oxidize, form gums, and foul equipment.
You’re absolutely right that these are the same molecules used today to make polyolefins like polyethylene and polypropylene. But in this context, they were considered contaminants—something that compromised the stability of the oil.
L.K.:
One small thing I liked—the red “Received” stamp. It feels almost ceremonial.
F.M.:
It was very deliberate. That stamp recorded the arrival of the memo in the purchasing department. It told everyone this letter was formally entered into the record.
These plants ran on documentation as much as they ran on coal and steam. Even routine correspondence had to be accounted for—especially when a salesman was making the rounds.
L.K.:
Do you think Novak left any impression on the staff here?
F.M.:
Probably more than he realized. Even if they never bought a single gallon of Deep Rock oil, he succeeded in getting them to discuss it, circulate the specs, and write it into the files.
That’s the thing about good salespeople—they’re not just pitching. They’re planting the seed that might sprout years later when someone asks, “Don’t we have a spec sheet on this somewhere?”

L.K.:
Frank, I came across this little sequence from 1963. It starts with a teletype saying Milwaukee Solvay Coke was sending a one-gallon sample of absorbent oil, and ends with a pink memo rejecting it because of emulsification time. Can you walk me through what’s happening here?
F.M.:
Sure—this is a textbook example of how carefully they vetted any new oil. Solvay wasn’t just an OEM—they operated coke plants themselves, so they sometimes marketed surplus fractions like absorbent oil to other producers.
That teletype was a formal notification: We’ve got a sample coming, and if it passes your tests, we’d be talking about 10,000 gallons. That’s a substantial volume—enough to supply operations for quite a while.
L.K.:
Why was emulsification such a big deal?
F.M.:
Absorbent oil has to separate cleanly from water after use. If you get a lot of emulsion—tiny water droplets suspended in the oil—it complicates regeneration and can cause fouling in the scrubbers or the stills.
Their spec required 95% separation in 10 minutes. The Milwaukee Solvay sample hit 95% only after more than 26 minutes. That was way too slow. Even if everything else looked good, they weren’t going to gamble on plugging equipment.
L.K.:
It struck me as interesting that another coke plant was pitching product to Interlake. Was that common?
*F.M.:
It wasn’t unusual at all. The big byproduct producers—Solvay, Koppers, and sometimes Wilputte—were always looking for outlets for surplus. If you were fractionating your own oils, you ended up with more than you could consume internally.
Selling to other plants was a smart way to monetize the excess. You’d see this especially when the market was soft or when one plant had temporarily high yields.
L.K.:
What do you make of the speed of this process? The sample arrived November 12th, and by the 18th they’d already issued a formal rejection.
F.M.:
That’s exactly how it worked. These were big operations with established lab protocols. As soon as the sample came in, they would test viscosity, sulfur content, flash point—and especially emulsification.
Once it failed that one parameter, the rest didn’t matter. Within a week, they closed the file.
L.K.:
I have to ask—do you think the sales rep at Solvay was disappointed, or was this just part of the game?
*F.M.:
Oh, he was probably disappointed. But any good industrial salesman knows you can’t win them all. Sometimes you’re pitching a product that’s surplus, or slightly out of spec, and you hope someone has more tolerance for it.
But you also know that even a rejection isn’t a dead end. You got your sample through the door. You got your name on record. You planted a seed that might pay off later—if the incumbent supplier faltered or if specs changed.
L.K.:
That’s one of the things I’ve learned from this folder—how much of running a plant was about saying no.
*F.M.:
Absolutely. Operations is 90% about consistency. Saying no to an unknown oil is just as important as saying yes to the right one.

L.K.:
Frank, I’m looking at these two sheets from 1962–63. One is a set of specifications for different oils, and the other is a breakdown of consumption and storage. Can you walk me through why they were tracking all this in such detail?
F.M.:
Absolutely. Fuel was a huge part of operating costs, and there was no room for improvisation. You had everything from Euclids and cranes running on diesel to stationary boilers and furnaces on heavier oils. Each application had strict requirements—viscosity, sulfur content, distillation range—because if you used the wrong grade, you’d foul injectors or burners or create dangerous deposits.
L.K.:
I see the specs track things like API gravity, pour point, sulfur percentage, viscosity, and flash point. Why so much detail?
F.M.:
Every one of those parameters affected either performance or safety. For example, the pour point told you whether the fuel would stay fluid in winter. Viscosity determined how well it atomized in burners. Sulfur content impacted corrosion and emissions. Even flash point mattered—especially in torch oil.
It wasn’t optional to meet these specs. Any new supplier had to certify conformance before the first delivery.
L.K.:
And what about the second sheet? That one lists the annual gallons, the cost per gallon, and the storage tanks.
F.M.:
That was the procurement ledger. You’re seeing exactly how much of each oil they bought in a year, from which supplier, and where it was stored. This allowed purchasing, accounting, and operations to reconcile inventory and plan budgets.
Notice that furnace oil accounted for over 700,000 gallons—that’s why it got special attention. Any interruption in that supply could shut down heating or coke production.
L.K.:
It looks like the prices varied quite a bit—some under 10 cents per gallon, others closer to 15 cents. What caused that spread?
*F.M.:
Mostly grade and delivery logistics. Torch oil was more refined, often smaller volume, so higher per-gallon cost. Bulk furnace oil could be cheaper per gallon but came in tank cars or large truckloads. And the source mattered too—some was from Standard Oil, some from American Oil, all with different contracts.
L.K.:
How did they handle quality assurance? Did the lab check every delivery?
*F.M.:
They spot-checked batches periodically. Incoming shipments were sampled for sulfur, water content, and viscosity. If a supplier ever failed spec, they were suspended from deliveries until it was corrected. That’s why you see “sweet” specified in almost every grade—they didn’t want high-sulfur product slipping in.
L.K.:
So this is basically the plant’s whole fuel economy in two sheets?
*F.M.:
That’s exactly right. You’re looking at the heart of how the plant stayed running. Without tight controls on specs and inventory, you risked either downtime or equipment damage.
It may look dry, but this is what kept the place on budget and operating day in, day out.
